|
Earlier this week, the U.S. National Academies published a sweeping report on the U.S.' role in ocean plastic pollution. You can read the full report here.
I was invited to write an op-ed style response, which I did with two of my colleagues. Today I saw our write-up was the feature story on The Conversation's front page!
1 Comment
The following was originally written for The Conversation, and republished in The San Francisco Chronicle. I'm re-posting here. Plastic pollution in the world’s oceans has become a global environmental crisis. Many people have seen images that seem to capture it, such as beaches carpeted with plastic trash or a seahorse gripping a cotton swab with its tail. As a scientist researching marine plastic pollution, I thought I had seen a lot. Then, early in 2017, I heard from Alex Weber, a junior at Carmel High School in California. Alex emailed me after reading my scientific work, which caught my eye, since very few high schoolers spend their time reading scientific articles. She was looking for guidance on an unusual environmental problem. While snorkeling in the Monterey Bay National Marine Sanctuary near the town of Carmel-by-the-Sea, Alex and her friend Jack Johnston had repeatedly come across large numbers of golf balls on the ocean floor. As environmentally conscious teens, they started removing golf balls from the water, one by one. By the time Alex contacted me, they had retrieved over 10,000 golf balls – more than half a ton. Golf balls sink, so they don’t become eyesores for future golfers and beachgoers. As a result, this issue had gone largely unnoticed. But Alex had stumbled across something big: a point source of marine debris – one that comes from a single, identifiable place – polluting federally protected waters. Our newly published study details the scope of this unexpected marine pollutant and some ways in which it could affect marine life. Cleaning up the mess Many popular golf courses dot the central California coast and use the ocean as a hazard or an out-of-bounds. The most famous course, Pebble Beach Golf Links, is site of the 2019 U.S. Open Championship. Alex wanted to create a lasting solution to this problem. I told her that the way to do it was to meticulously plan and systematically record all future golf ball collections. Our goal was to produce a peer-reviewed scientific paper documenting the scope of the problem, and to propose a plan of action for golf courses to address it. Alex, her friends, and her father paddled, dove, heaved and hauled. By mid-2018 the results were startling: They had collected nearly 40,000 golf balls from three sites near coastal golf courses: Cypress Point, Pebble Beach and the Carmel River Mouth. And following Alex’s encouragement, Pebble Beach employees started to retrieve golf balls from beaches next to their course, amassing more than 10,000 additional balls. In total, we collected 50,681 golf balls from the shoreline and shallow waters. This represented roughly 2.5 tons of debris – approximately the weight of a pickup truck. By multiplying the average number of balls lost per round played (1-3) and the average number of rounds played annually at Pebble Beach, we estimated that patrons at these popular courses may lose over 100,000 balls per year to the surrounding environment. The toxicity of golf balls Modern golf balls are made of a polyurethane elastomer shell and a synthetic rubber core. Manufacturers add zinc oxide, zinc acrylate, and benzoyl peroxide to the solid core for flexibility and durability. These substances are also acutely toxic to marine life. When golf balls are hit into the ocean, they immediately sink to the bottom. No ill effects on local wildlife have been documented to date from exposure to golf balls. But as the balls degrade and fragment at sea, they may leach chemicals and microplastics into the water or sediments. Moreover, if the balls break into small fragments, fish, birds or other animals could ingest them. The majority of the balls we collected showed only light wear. Some could even have been resold and played. However, others were severely degraded and fragmented by the persistent mechanical action of breaking waves and unremitting swell in the dynamic intertidal and nearshore environments. We estimated that over 60 pounds of irrecoverable microplastic had been shed from the balls we collected. Game-changer
Thanks to Alex Weber, we now know that golf balls erode at sea over time, producing dangerous microplastics. Recovering the balls soon after they are hit into the ocean is one way to mitigate their impacts. Initially, golf course managers were surprised by our findings, but now they are working with the Monterey Bay National Marine Sanctuary to address the problem. Alex is also working with managers at the sanctuary to develop cleanup procedures that can prevent golf ball pollution in these waters from ever reaching these levels again. Although her study was local, her findings are worrisome for other regions with coastal golf courses. Nonetheless, they send a positive message: If a high school student can accomplish this much through relentless hard work and dedication, anyone can. Jostling us back and forth like rag dolls, we navigated our rented Jeep Wrangler along the rutted, unpaved roads of rural Lanai. I was visiting my friend Matt Koller, who was working on whale watching boats in Maui, when we decided to take a day trip to the Pineapple Isle of Lanai. Though only a 45-minute ferry ride from Maui, Lanai is easily accessible, but a world apart. Sparsely populated, it has only 3,000 permanent residents, compared to Maui’s 150,000. We had come here in search of the beautiful, unspoiled Hawaii we had read about in books. And by traveling to Shipwreck Beach – one of the most remote beaches on Lanai – we were sure to find it. Or so we thought. It had been roughly 45 minutes since we last saw pavement when we reached the beach at the road’s northern terminus. We were stunned into silence by what we found. We found trash. Trash everywhere. The majority – if not the entirety – of this trash was not left here from the beach’s rare visitors, but rather had washed up from places near and far.  Some examples of the debris found on Henderson Island. From: Lavers and Bond. 2017. PNAS 114(23): 6052-55. Some examples of the debris found on Henderson Island. From: Lavers and Bond. 2017. PNAS 114(23): 6052-55. This may have been both the most remote and the most trashed beach I had ever visited, and considering I grew up on an island known for its now-defunct garbage dump (which could be seen from outer space), that’s something I don’t say lightly. Sadly, encountering dense concentrations of debris on remote beaches is not an experience unique to us. Earlier in 2017, a paper was published that made national headlines and caught my eye. This happenstance study occurred when two seabird researchers visited Henderson Island in the south Pacific to survey nesting seabirds. Henderson Island is uninhabited by people; the nearest human settlement is Pitcairn Island, approximately 100 miles to the west, and even that island has less than 100 inhabitants. It is not hyperbole to call Henderson Island one of the most remote islands on earth. Counterintuitively, when the seabird researchers landed here in mid-2015, what they found astonished them in a way similar to how I had been shocked on Lanai. They recorded the highest density of debris of any beach on the planet, in some places as dense as 700 pieces per square meter. That’s 700 bottle caps, straws, plastic fragments, and other refuse in an area approximately the size of your beach towel. While I was standing on that beach in Lanai – with pieces of sand and plastic between my toes – I thought of Henderson Island and had a revelation. Popular beaches that are devoid of obvious litter (on Maui, for example) aren’t as naturally pristine as visiting tourists may believe--they’re cleaned. Veritable armies of government and hospitality workers maintain, manicure, and clean the tourist beaches of Florida, Hawaii, the Caribbean, the Mediterranean and beyond to keep them aesthetically pleasing. Conversely, remote beaches, like Shipwreck Beach on Lanai, are hard to access and don’t have the tourist demand to be kept clean. In some regions, even some extremely remote regions, these beaches become natural landfills. I call this the paradox of “pristine” beaches. Earlier this year while visiting the Galapagos, I encountered another example of this phenomenon. On the frequently visited beaches near the town of Puerto Ayora on Santa Cruz Island, almost no litter was to be found, while on the more remote beaches of nearby Floreana Island the sand was peppered with small, yet obvious, plastic fragments. Santa Cruz Island has more than one hundred times the human population of Floreana Island, but I saw far more anthropogenic debris on Floreana’s beaches. Standing on Shipwreck Beach in Lanai and Floreana Island in the Galapagos, and staring not only at the ocean, but also at the technicolor trash that had washed up made me wonder: do truly pristine beaches still exist?
In our plastic age, it seems highly unlikely. But documenting and cleaning the beaches is a good first step at remedying the mess we have made, and the best part is anyone can do it. The next time you’re at a beach, pick up a few pieces of trash if you find any--the planet and your fellow humans will be grateful. It’s the least we can do. The following - originally written for The Conversation, and republished in The Washington Post - is a layperson summary of the third chapter of my dissertation. I'm re-posting here. As you bite down into a delicious piece of fish, you probably don’t think about what the fish itself ate – but perhaps you should. Over 50 species of fish have been found to consume plastic trash at sea. This is bad news, not only for fish but potentially for humans who rely on fish for sustenance. Fish don’t usually die as a direct result of feeding on the enormous quantities of plastic trash floating in the oceans. But that doesn’t mean it’s not harmful for them. Some negative effects that scientists have discovered when fish consume plastic include reduced activity rates and weakened schooling behavior, as well as compromised liver function. Most distressingly for people, toxic compounds such as PBDEs that are associated with plastic transfer to and bioaccumulate in fish tissues. This finding is troubling because it means these toxic substances could further bioaccumulate in us if we consume fish that have eaten plastic. Numerous species sold for human consumption, including mackerel, striped bass and Pacific oysters have been found with these toxic plastics in their stomachs too. It is well-known that our plastic trash poses a serious threat to marine animals, but we are still trying to understand why animals eat it. Typically, research has concluded that marine animals visually mistake plastic for food. While this may be true, the full story is likely more complex. For example, in a recent study with colleagues at the University of California, Davis, we showed that plastic debris may also smell attractive to marine organisms. That study focused on seabirds, but now my co-authors and I have found that plastic trash has a similar effect on anchovies – a critical part of ocean food chains.  Filming an experimental school of anchovy at the Aquarium of the Bay Filming an experimental school of anchovy at the Aquarium of the Bay Sniffing out the role of smell Olfaction (smell) is a very important sense for marine animals, including fish. Sharks can smell minute quantities of blood over long distances, which helps them find prey. And scientists believe that salmon’s sense of smell helps them navigate up rivers to the specific tributaries where they were born to spawn. Fish may use their sense of smell in behavioral contexts including mating, homing, migrating and foraging. We tested the idea that plastic debris might smell attractive to the Northern anchovy (Engraulis mordax), a common schooling fish found off the West Coast of North America. Known as forage fish, anchovies are critically important species ecologically and economically. Unfortunately, they have also been found to eat plastic in the wild. Working with anchovies is challenging because they require very specific water conditions and school size to behave normally. They need to be in cold, fast-flowing water in schools of at least 100 individuals. When that happens, the anchovies display their contentment by swimming slowly and directly into the flow of water – a behavior known as positive rheotaxis. Luckily, we were able to collaborate with the Aquarium of the Bay in San Francisco, where they have expertise in keeping these fish happy and healthy. Our olfactory experiment When we started the experiment we did not know whether adult anchovies used their sense of smell to find food at all, let alone whether smell might lead them to consume plastic. To test our hypothesis that it would, we soaked krill (tiny shrimp-like crustaceans that anchovies eat) or plastic debris and clean plastic in seawater for several hours, allowing the water to take on the smell of the material steeping in it. We then filtered our krill or plastic “tea,” presented it to the anchovy schools, and observed their behavior. When fish are searching for food in groups, their behavior changes in predictable ways: They clump together near the interesting stimulus and dart around, altering their body position relative to the water current. To compare how anchovies responded to the scents of krill and plastic, we hung a specially designed apparatus with a GoPro camera attached over their tank to film the school’s behavior from above. In addition to analyzing what anchovies did when they detected these odors, we also filmed their anchovies’ behavior while feeding on krill and when they were presented with control treatments of unscented seawater. This gave us baseline information about the schools’ behavior, which we could compare to their responses when they were presented with the different odors. Using a combination of automated computer analyses and diligent observer scoring, we evaluated how tightly the schools clumped together and how each fish’s body positioning relative to the direction of water flow changed before and after adding an odor solution to the tank. As we predicted, when the anchovies were feeding, schools became more densely clumped and changed their body positioning so that instead of all fish facing directly into the oncoming current, their bodies aligned more haphazardly as they searched for food morsels. In the control treatments, with no food or food odors present, we did not observe these changes. When we injected seawater scented with krill into the tank, the anchovies responded as if they were searching for food – which in this case was not there. And, importantly, when we presented them with seawater scented with odors of plastic debris, the schools responded in nearly the same way, clumping together and moving erratically as they would if they were searching for food. This reaction provided the first behavioral evidence that a marine vertebrate may be tricked into consuming plastic because of the way it smells. Reducing plastic pollution
This research confirms several things. First, we showed that Northern anchovies use odors to locate food. This may sound intuitive, but before we did this study there was scant behavioral evidence that adult forage fish, such as anchovies, sardines and herring used smell to find food. Our main finding was that plastic debris is likely confusing for marine consumers because of both its appearance and its smell. That’s a problem, because if plastic looks and smells interesting to fish, it will be very hard for them to discern that is it not food. This study also suggests that our consume-and-dispose culture is coming back to haunt us via the fish we eat. The next big question that it raises is whether plastic-derived contaminants can be transferred from plastic-eating fish to fish-eating humans. One way to mitigate the problem is to figure out why animals confuse plastic for prey so frequently, and our research has helped to do that. However, everyone can do something right now about ocean plastic pollution by avoiding single-use plastic items and recycling plastic upon disposal. There is more work to be done, but we know enough now to make substantial headway on this global environmental issue.  Wisdom attending to her chick on Midway Atoll in 2013. Photo by: USFWS Wisdom attending to her chick on Midway Atoll in 2013. Photo by: USFWS Nature’s superlatives are inherently fascinating. The fastest. The biggest. The smallest. The strongest. The tallest. This post is about another one of those superlatives: the oldest. Specifically, this post is about the oldest vertebrates. Many of the oldest vertebrates are marine creatures, a happy coincidence given my expertise. For reference, the life expectancy of any given person living in the United States is approximately 79. In the animal kingdom that’s pretty old – only a tiny fraction of non-human animals regularly live that long (think tortoises, whales, elephants). But again, we’re talking about superlatives; so how old can humans get? The oldest person on record was Jeanne Calment, who died at the ripe old age of 122 in 1997. Demographers estimate the maximum human lifespan to be 125, based on mortality data. But if you think that’s old, we’re just getting started. The oldest non-human vertebrates are much older indeed. Let’s start with birds, some of my favorite organisms. There are records of captive flamingos, condors, parrots, and cranes living into their eighties, but no wild bird is known to live that long. However, this brings up an important issue in gerontology: how do we verify the age of wild animals? The most intuitive way is to identify or label an individual with a unique mark so that scientists can track the individual over time. The most well known way of doing this is bird banding, where a metal band with a unique number is affixed to a bird’s leg, like an ankle bracelet of sorts. But to get age information on a banded bird you need two things: to know roughly how old the bird is when it is banded and to recapture the bird at some later date. These are both difficult endeavors. Birds aren't easy to age: while many birds sport distinct juvenile and adult plumages, once the bird reaches adult plumage there is often no way to tell exactly how old the individual is; birds’ feathers don’t gray like human hair does. Recapturing a banded bird at some later date is even more of a challenge: banded birds are almost never recaptured. For example, the recapture rate of previously banded songbirds is a minuscule 1.3%. Seabirds have much higher recapture rates, however, due to their breeding site fidelity. If conditions are right, most seabirds return to the same island – and often the exact same location on the island – annually to breed. Seabirds also happen to have long lifespans; some albatross don’t even start breeding until they are nearly 10 years old. The oldest known seabird is also the oldest known wild bird on the planet. She is a Laysan Albatross named Wisdom and she is at least 66 years old. Wisdom was banded as an adult in 1956 on Midway Atoll in the north Pacific by US Fish and Wildlife Service biologist Chan Robbins. Even more remarkable: she’s still reproducing; earlier this year she hatched her 41st chick! By comparison, the oldest human to conceive and give birth naturally was 59. Through her spectacular longevity and long reproductive history, Wisdom has become possibly the most famous wild bird in the world; there have been books written about her and she even has her own Facebook page (as a funny aside, we actually share 11 friends in common)! As far as albatross go, Laysan Albatross like Wisdom are some of the smallest, and in longevity, size matters. In general, the larger the species, the longer lifespan. It is, therefore, interesting to ponder how old some of the great albatross wandering the Southern Ocean might be. These flying giants are about twice the size of a Laysan Albatross and have been recorded to live longer than 50 years, but we simply don’t know what their maximum lifespan is. My guess is a good deal older than 60, possibly older than 70. Only time and data will tell. If larger animals tend to have longer lifespans, then baleen whales, the largest animals on earth, should be really old. This proves true for the Bowhead Whale, a large arctic filter feeder. They are enormous, weighing up to 100 tons (200,000 pounds), making them the second heaviest animal on the planet behind the Blue Whale. The first hints at the Bowhead Whale’s extreme longevity came about in an unusual way – modern Inuits discovered harpoons from 19th century hunters in the bodies of whales they captured. Simple arithmetic ages those whales at over 100 years old.  The Greenland Shark's 5-minutes of fame, on the cover of Science magazine in August 2016. The Greenland Shark's 5-minutes of fame, on the cover of Science magazine in August 2016. For bowheads that do not have harpoon fragments lodged in their body, scientists have used a technique called aspartic acid racemization to age individuals. The basic premise is this: aspartic acid is an amino acid (a building block of proteins) that exists in two different forms called D and L enantiomers, which are essentially mirror images of each other. Living tissue produces only the L, but not D, enantiomer. In metabolically inactive tissue, such as the lens of the eye, the L enantiomers slowly convert to D enantiomers – a process called racemization. The ratio can be used to age metabolically inactive tissue, and thus age the individual. Of course, analysis of the eye lens tissue requires lethal sampling: the animals must be dead for the sample to be taken. Luckily for scientists, Inuit hunters agreed to donate Bowhead Whale eyes for research. The results of the aspartic acid racemization were stunning: 4 of the 24 males tested (17%) were over 100 years old. One special individual, 95WW5, was estimated to be 211 years old, by far the oldest mammal on record. To put that into perspective, this whale was born around the time The Bill of Rights was ratified. Recently however, the Bowhead Whale was dethroned as the longest-lived vertebrate by a massive cold-water fish, the Greenland Shark. These mild-mannered sharks are so slow and strange-looking their Latin name – Somniosus microcephalus – translates to “sleepy and small headed”. They are cold-blooded and live in frigid arctic waters year round. They grow exceptionally slowly – less than 1 cm per year, yet can grow to over 15 feet in length – and have an extremely slow metabolic rate; this allows them to live a very long time. But even the scientists that study them were shocked to find out just how old these gentle giants can be. To determine the age of these sharks, the researchers used a technique called bomb radiocarbon dating. The process works as follows: most carbon atoms – nearly 99% of them – have 6 neutrons, but one-in-a-trillion carbon atoms has 8 neutrons. These different forms of carbon are known as the isotopes carbon-12 and carbon-14. As a result of the detonation of hundreds of atomic weapons in the 1950s, carbon-14 in the atmosphere doubled in the late 1950s and 1960s, and has been tapering off ever since. Because living organisms assimilate carbon from the environment, that doubling of carbon-14 in the atmosphere is reflected in the tissues and cells of all organisms that lived during the period of nuclear testing. Scientists studying the Greenland Shark used differing quantities of “bomb pulse” carbon-14 in each individual to obtain an age estimate. There is a negative relationship between percentage of carbon-14 and age. In other words, the less carbon-14, the older the animal, because more of its tissues would have formed prior to nuclear testing and thus have incorporated more pre-bomb carbon-12. Using these methods, the authors calculated that the oldest shark in their sample was 392 years old, with a potential error of 120 years. This means that even the lowest estimate of its age (272) still beats out Bowhead Whale 95WW5 as the oldest vertebrate by over 60 years! Most likely this shark was around 400 years old, born during the time of Shakespeare and Galileo. At the upper end of the estimate, this individual could have been 500 years old, four times as old as the maximum human lifespan. These findings were so groundbreaking it was covered by nearly 200 news outlets worldwide and it landed the Greenland Shark on the cover of Science, one of the most prestigious scientific publications in the world. Another remarkable finding was that Greenland Sharks don’t become sexually mature until at least 150 years old. This is by far the most delayed sexual maturation of any animal known. Their extremely long lifespan and extraordinarily slow reproduction also means they are vulnerable to exploitation and disturbance. If their population were reduced by some anthropogenic or natural perturbation, it would take them centuries to recover, if they recover at all. For this reason, the longest-lived organisms on the planet are also some of the most imperiled by human activities. The lesson is that we are still learning about the world around us, especially in the ocean’s depths. As a result, we need to be especially cautious when taking actions that affect the natural world. If we’re not careful, we could wipe out the most fascinating species before we can unlock the mysteries they’re holding.
Imagine that you are constantly eating, but slowly starving to death. Hundreds of species of marine mammals, fish, birds, and sea turtles face this risk every day when they mistake plastic debris for food. Plastic debris can be found in oceans around the world. Scientists have estimated that there are over five trillion pieces of plastic weighing more than a quarter of a million tons floating at sea globally. Most of this plastic debris comes from sources on land and ends up in oceans and bays due largely to poor waste management. Plastic does not biodegrade, but at sea large pieces of plastic break down into increasingly smaller fragments that are easy for animals to consume. Nothing good comes to animals that mistake plastic for a meal. They may suffer from malnutrition, intestinal blockage, or slow poisoning from chemicals in or attached to the plastic. Despite the pervasiveness and severity of this problem, scientists still do not fully understand why so many marine animals make this mistake in the first place. It has been commonly assumed, but rarely tested, that seabirds eat plastic debris because it looks like the birds’ natural prey. However, in a study that my coauthors and I just published, we propose a new explanation: For many imperiled species, marine plastic debris also produces an odor that the birds associate with food. A nose for sulfur Perhaps the most severely impacted animals are tube-nosed seabirds, a group that includes albatrosses, shearwaters and petrels. These birds are pelagic: they often remain at sea for years at a time, searching for food over hundreds or thousands of square kilometers of open ocean, visiting land only to breed and rear their young. Many are also at risk of extinction. According to the International Union for the Conservation of Nature, nearly half of the approximately 120 species of tube-nosed seabirds are either threatened, endangered or critically endangered. Although there are many fish in the sea, areas that reliably contain food are very patchy. In other words, tube-nosed seabirds are searching for a “needle in a haystack” when they forage. They may be searching for fish, squid, krill or other items, and it is possible that plastic debris visually resembles these prey. But we believe that tells only part of a more complex story. Pioneering research by Dr. Thomas Grubb Jr. in the early 1970s showed that tube-nosed seabirds use their powerful sense of smell, or olfaction, to find food effectively, even when heavy fog obscures their vision. Two decades later, Dr. Gabrielle Nevitt and colleagues found that certain species of tube-nosed seabirds are attracted to dimethyl sulfide (DMS), a natural scented sulfur compound. DMS comes from marine algae, which produce a related chemical called DMSP inside their cells. When those cells are damaged – for example, when algae die, or when marine grazers like krill eat it – DMSP breaks down, producing DMS. The smell of DMS alerts seabirds that food is nearby – not the algae, but the krill that are consuming the algae. Dr. Nevitt and I wondered whether these seabirds were being tricked into consuming marine plastic debris because of the way it smelled. To test this idea, my coauthors and I created a database collecting every study we could find that recorded plastic ingestion by tube-nosed seabirds over the past 50 years. This database contained information from over 20,000 birds of more than 70 species. It showed that species of birds that use DMS as a foraging cue eat plastic nearly six times as frequently as species that are not attracted to the smell of DMS while foraging. To further test our theory, we needed to analyze how marine plastic debris smells. To do so, I took beads of the three most common types of floating plastic – polypropylene and low- and high-density polyethylene – and sewed them inside custom mesh bags, which we attached to two buoys off of California’s central coast. We hypothesized that algae would coat the plastic at sea, a process known as biofouling, and produce DMS. After the plastic had been immersed for about a month at sea, I retrieved it and brought it to a lab that is not usually a stop for marine scientists: the Robert Mondavi Institute for Food and Wine Science at UC Davis. There we used a gas chromatograph, specifically built to detect sulfur odors in wine, beer and other food products, to measure the chemical signature of our experimental marine debris. Sulfur compounds have a very distinct odor; to humans they smell like rotten eggs or decaying seaweed on the beach, but to some species of seabirds DMS smells delicious!
Sure enough, every sample of plastic we collected was coated with algae and had substantial amounts of DMS associated with it. We found levels of DMS that were higher than normal background concentrations in the environment, and well above levels that tube-nosed seabirds can detect and use to find food. These results provide the first evidence that, in addition to looking like food, plastic debris may also confuse seabirds that hunt by smell. When trash becomes bait Our findings have important implications. First, they suggest that plastic debris may be a more insidious threat to marine life than we previously believed. If plastic looks and smells like food, it is more likely to be mistaken for prey than if it just looks like food. Second, we found through data analysis that small, secretive burrow-nesting seabirds, such as prions, storm petrels, and shearwaters, are more likely to confuse plastic for food than their more charismatic, surface-nesting relatives such as albatrosses. This difference matters because populations of hard-to-observe burrow-nesting seabirds are more difficult to count than surface-nesting species, so they often are not surveyed as closely. Therefore, we recommend increased monitoring of these less charismatic species that may be at greater risk of plastic ingestion. Finally, our results provide a deeper understanding for why certain marine organisms are inexorably trapped into mistaking plastic for food. The patterns we found in birds should also be investigated in other groups of species, like fish or sea turtles. Reducing marine plastic pollution is a long-term, large-scale challenge, but figuring out why some species continue to mistake plastic for food is the first step toward finding ways to protect them. If you know me, you know I am passionate about science communication. Not surprisingly, I was overjoyed when so many fantastic science communicators wanted to write pieces about the paper we recently published in Science Advances. If you're looking for coverage that I believe to be especially compelling, I point you in the direction of the stories by NPR (as heard on All Things Considered), Audubon, and The Atlantic.
Also, be sure to check out the popular science write up I did myself in The Conversation! Empathy is the capacity to understand or feel what another being is experiencing from within the other's frame of reference. Simply put, empathy is the ability to place yourself in another’s shoes. Despite learning the golden rule prior to elementary school, people often behave selfishly, disregarding the consequences of their words and actions on others. I can be as guilty of this as anyone. For most, myself included, empathy does not come naturally. However, by persevering through hardships along with serious introspection, we can put ourselves in another’s shoes, feel what they feel, and in doing so, feel empathy.
Empathy is a powerful tool. Empathetic understanding deepens emotional relationships with others. It can make you a better co-worker, partner, and friend. Yet despite the powers of empathetic thinking and action, our species treats the natural world with a disheartening lack of empathy, which hinders not only our conservation of the natural world, but our understanding of ecology and animal behavior. In 1909, German biologist Jakob von Uexküll published a book where he coined the term “umwelt,” to describe the sensory world in which an organism exists and acts as a subject. This term is used to succinctly describe the total sensory experience of an individual. For a sensory ecologist, understanding the umwelt of the organisms you’re studying is imperative; if you use a human frame of reference to design experiments for a non-human animal, the results you record and inferences you make will likely be misleading. To think about this in more detail, consider the example of a dog whistle. High pitched dog whistles are used to train dogs to perform certain tasks. The upper limit of human hearing is 17-20 kHz; a dog whistle emits its shrill tone at 23-54 kHz. To human ears, a dog whistle sounds like a quiet hissing, certainly nothing unpleasant. By comparison, your pet dog can hear up 45kHz. So that dog whistle, though benign to you, sounds worse than nails on a chalkboard for Fido. The reason a dog whistle is such an effective training tool is due to the difference in hearing capabilities (auditory umwelt) between humans and canines. And by employing the concept of umwelt to imagine how unpleasant a dog whistle sounds as a dog, you may be opposed to using one to train a dog in the future. Herein lies the connection of umwelt and empathy. Understanding the sensory world of another organism can help to understand its experience despite having never shared the same experience. Umwelt not only differs between species, but between individuals within a species. Staying with the example of hearing, if I (a 28-year-old) go for a walk in the park with my 85-year-old grandmother, I will hear more bird song than she will because humans lose the ability to hear higher frequencies as they age. If I organize songbird surveys with retired volunteers, I need to be aware of and adjust for the volunteers' umwelt (perhaps using visual rather than auditory surveys). I can also empathize by accepting their limitations and abilities while describing the songs that are inaudible to them. Despite living in the same world, we all inhabit different subjective realities – we interpret the world and our experiences differently. Harnessing the diversity of human experiences gives us collective strength. Applying the umwelt concept gives us a greater ability to understand the sensory world and experiences of non-human organisms. If we can grapple with these differences between people as well as between species, we can foster more supportive communities and better conserve the natural world. 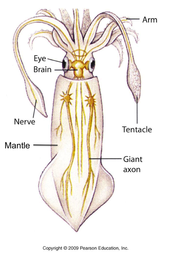 Basic neuroanatomy of a squid. Basic neuroanatomy of a squid. Danish Nobel-laureate August Krough once said in what is now known as the Krough Principle, "for [many] problems there will be some animal of choice, on which it can be most conveniently studied." This means that if you are interested in studying a certain trait, such as a nose’s ability to smell, the organism to study it on should have a very exaggerated form of that trait. An example of this in neurobiology is the squid and its giant axon. These cephalopods developed a giant axon as a way to relay sensory information through their elongated mantle to their brain as instantaneously as possible. For this reason, squid are often the animal model used in basic research on neurons with implications for human health. To understand the sense of smell (olfaction), it seems reasonable that studying the animal with the world’s largest nose would be a good place to start. So which organism has the largest nose in the animal kingdom? While Cyrano de Bergerac or an elephant would be good guesses, by most traditional measures, the Sperm Whale (Physeter macrocephalus) takes the cake. The Sperm Whale is the largest toothed predator to ever live, growing to a maximum length of 20 meters (65 feet) and weight of 60 tons (120,000 pounds)! Despite these colossal dimensions, their most striking physical feature is their huge, blocky head. Heck, their species’ name, macrocephalus, means “large head.” This unusual noggin is mostly nose and neighboring structures. The Sperm Whale’s nose accounts for roughly a third of the animal’s total length and weight. This led Danish physiologist Bertel Møhl to quip that a Sperm Whale is basically just a nose with an outboard motor. With such a tremendous nose, the Sperm Whale must possess an amazing sense of smell, right? Wrong. All odontocetes (the toothed whales; e.g., dolphins, Orcas, beaked whales, etc.) including Sperm Whales have no olfactory bulb, the region of the vertebrate brain essential for transmitting and deciphering olfactory information. In other words, they are believed to have no sense of smell whatsoever! So what is this massive nose used for? In 1985 Sperm Whales were given full international protection from hunting, but for the two centuries prior, whalers killed over one million Sperm Whales, reducing their global population by nearly 70%. What made Sperm Whales so highly sought after was not their meat or blubber, but their spermaceti. Approximately 5000 liters (over 1000 gallons) of this waxy substance can be found in an adult male Sperm Whale’s head, and once processed it can be used for lubrication, leatherworking, lipstick, and lantern fuel. Spermaceti in liquid form closely resembles… well, male ejaculate of course. It’s how the Sperm Whale got its common name. Like other toothed whales, the Sperm Whale finds its prey by echolocation, sending out an intense beam of high-frequency sound, listening to the returning echoes to locate prey at depths of 200-2,000 meters. However, the spermaceti organ is unique to Sperm Whales, but why? The reason is because spermaceti has an incredible resonating capability. Here’s how it works: a Sperm Whale produces an echolocation click near its blowhole; counterintuitively, the sound travels backwards towards the base of the head, through the spermaceti organ. At the base of the head, the sound is reflected off an airsac and focused out through the front of the whale’s head. This circuitous route helps make the sound as loud and directionally focused as possible. Measured at 230dB underwater – equivalent to 170dB on land (as loud as a gunshot three feet away) – the Sperm Whale’s echolocation click is the loudest biologically produced sound on earth and it wouldn’t be possible without their unique spermaceti organ. So there you have it, the largest nose in the animal kingdom is actually the world’s most powerful biological resonating chamber.
(Side note: how does the giant squid, the Sperm Whale’s favorite prey, stand any chance against these whales, capable of detecting squid over 500 meters away in nearly complete darkness? Read my post from last October to find out.) The Sperm Whale is an auditory wonder, fine-tuned over millions of years of evolution. To protect these amazing creatures into perpetuity, society needs to consider how we are impacting Sperm Whales today. Anthropogenic noise pollution from shipping traffic, naval operations, and oil exploration negatively affects a myriad of oceanic life including marine fish, baleen whales, and the aforementioned cephalopods. However, the consequences may be gravest for the toothed whales. For example, scientists have evidence to suggest that beaked whales quickly dive to and from great depths to evade loud sounds, giving them decompression sickness – known by its more familiar name, the bends – which may lead to stranding. It has also been shown that Sperm whales avoid anthropogenic noise, which has been implicated in their stranding. It is time to use our knowledge of sensory biology for good, developing mitigation strategies that protect toothed whales from pervasive noise pollution. If we do so, future generations can continue to marvel at the world’s most impressive noses that cannot smell a thing. Last week, Grant Humphries told me about a fantastic new piece of research produced by the marine debris group from CSIRO's Ocean & Atmosphere flagship, based in Hobart, Tasmania. The researchers reported that they were able to detect a signature of plastic debris ingestion in several species of seabird by sampling the bird's preen gland oil and analyzing that sample on a GC-MS instrument. A GC-MS is able to detect and identify compounds at trace-level concentrations; the compounds that the researchers were interested in detecting were three common plasticizers, called phthalates.
This is awesome by itself, but the real breakthrough is that previously the only way to determine if a live seabird had ingested plastic was to lavage its stomach and sift through the regurgitated contents for plastic pieces. There are numerous problems with this approach; it would only show if the bird recently ingested plastic, data may be biased toward sick or weak individuals, and it's extremely invasive. This new GC-MS method could represent a paradigm shift in how wildlife managers and conservation biologists screen populations of marine animals - not only birds - for plastic ingestion. Without further ado, here are the next five amazing things I learned while participating in Lund University's Sensory Ecology Course. 5. Through the eyes of a fly Insect compound eyes are some of the most fascinating and beautiful structures in the animal kingdom, but what does the world look like to them? For all my life, I thought these animals saw the world as a honeycomb. However, Dan-Eric Nilsson shocked me when he told our class that is almost certainly not the way insects perceive the world. Insects are often quite near-sighted, but we have no reason to believe they see the world in honeycomb fashion. All existing evidence points to the fact that their eyes form just one image (albeit, a blurry one), as we do. Did that surprise anyone else, or just me? 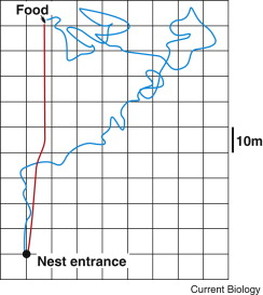 Generalized foraging path of a Cataglyphis ant (Figure from Willis 2012). Generalized foraging path of a Cataglyphis ant (Figure from Willis 2012). 4. Homeward bound – path integration in desert ants A remarkable example of homing behavior comes from a group of unassuming animals, the Saharan Desert Ants (Cataglyphis sp.). These ants leave their subterranean burrows during the day and wander an enormous distance (relative to their size) around the scorching Sahara Desert in search of food scattered randomly on the landscape. As a result, their path while searching for food is lengthy and meandering relative to their return trip, which is essentially a beeline back to the cool safety of their burrow (see figure on right). How do these tiny animals do this in the open desert with no physical or chemical landmarks (i.e., they do not follow a scent trail like other ant species)? They need to integrate two types of information to make the direct journey home: relative angle and distance traveled. In a landmark study published in 1981, researchers found that these ants use celestial cues to determine what angle to travel back home. Check out the video below. It wasn’t for another 25 years that scientists finally settled how these ants judge the approximate distance they need to travel. Researchers did this by first training Cataglyphis ants to walk from a burrow to a feeding station in an experimental arena. Then, the researchers caught the ants and experimentally manipulated the stride length in some ants to longer strides by attaching miniature stilts (pig bristles) to the ants' legs, made another group of ants’ strides shorter by cutting off the lower half of their legs, and left a third group unmanipulated as a control. Sure enough, the group with longer legs (and thus, longer stride-length) walked right past their burrow, while those with shorter legs walked only part of the way back before beginning a fruitless search for their burrow. The unmanipulated control ants made it back to their burrows perfectly. Finally, after a quarter century, the mystery had been solved; these ants use an onboard pedometer to judge distance, amazing! Read the article here and check out the NPR video summarizing the study below. 3. Pollution stinks Air pollution has negative effects on humans and animals, no surprise there. What is surprising however, are some of the ways in which air pollution can adversely affect human health. Researchers in Mexico have found that chronic exposure to the air pollution of Mexico City reduces people’s olfactory abilities and trigeminal nerve sensitivity. Individuals tested in Mexico City had a higher detection threshold and more difficultly discriminating between everyday odorants (e.g. coffee, orange drink, horchata) than people living in the nearby, less-polluted state of Tlaxcala. Even more alarming is that people living in Mexico City also had a worse detection threshold and discrimination ability of contaminated food odors (e.g. spoiled milk), which could lead to food poisoning or other food-borne illnesses. Further experiments testing how chronic, non-occupational exposure (i.e., non-miners) to airborne manganese in the mining region of Molango, Mexico showed similar negative effects on olfactory performance and trigeminal nerve sensitivity. This indicates reduced trigeminal nerve function, which could be an early warning sign of neurological damage because unlike other toxic metals, manganese is transported transynaptically to structures deep within the brain. Global regulations need to be enacted on these air pollutants before overwhelming adverse effects on human health become commonplace. 2. Electric feel – bees' electric sense Bees are remarkably resourceful little creatures. In 1973, Karl von Frisch won a Nobel Prize for decoding the honey bee’s waggle dance used by a returning forager to alert other bees in the hive to the relative angle (to the sun) and approximate distance to a food source, such as a patch of flowers. However, once a bee – alerted by the waggle dance of a hivemate – arrives at the flower patch, how does it choose which flower(s) to visit? Daniel Robert and colleagues at the University of Bristol recently discovered that they might select flowers to visit based on the flower’s electro-static charge. The theory goes like this: when bees fly through the air, they accumulate a positive charge, similar to what happens to a flying airplane. Since the flowers are grounded and have a slightly negative charge, when a positively-charged bee lands on a negatively-charged flower, some of the bees' positive charge is transferred to the flower. When other bees visit that same flower in the near future, they can detect the higher charge of the just-visited flower (relative to unvisited flowers) and choose to avoid it, since it would have less nectar than an unvisited flower. These are the first insects shown to have and potentially use their electric sense. This means that bees may integrate at least four different kinds of information (vision, olfaction, social, electrical) while foraging; multi-modal foraging at it’s best!  Namib Desert Golden Mole. Photo by G. Rathburn. Namib Desert Golden Mole. Photo by G. Rathburn. 1. Good vibrations – seismic communication in vertebrates Of all sensory modalities, I find the ones humans either don't possess (magnetoception, electroreception, etc.) or have very limited abilities in (e.g. olfaction) most fascinating. Last year, Robert Raguso gave a lecture at UC Davis where he described these signals as an invisible language just waiting to be decoded. Another example of this is substrate-transmitted seismic signals, which are imperceptible to us, but very important for animals specialized to detect them. Peter Narins has made a career investigating seismic communication. His work was the first to show that a vertebrate (male Gunther's White-lipped Frogs, Leptodactylus albilabris) incorporated ground-transmitted seismic signals in their display call, which unlike the auditory portion of their call, is used for male-male communication. Probably the most adorable example of an organism using seismic signals is the Namib Desert Golden Mole (Eremitalpa granti namibensis). These small, mammals are functionally blind and when they emerge from their burrows at night to forage, they literally swim through sand (see video below). Until Narins' group investigated the problem, no one was knew for sure why these animals foraged this way. It was found that the moles can actually detect vibrational differences produced by wind passing through mounds of dune grass, which is where the moles find their termite prey. Additionally, the researchers looked into the strange inner ear morphology of these animals and discovered they have the largest malleus (relative to their body size) of any animal known to science, which they believe is used to detect subtle vibrational changes in the substrate. This amazing feat of bioengineering is now being used to develop even more advanced earthquake detection systems. So if you ever find yourself asking, “why are we funding basic science, like that on the seismic sense of the golden mole?” One reason is because basic science can become applied science in the blink of an eye. Another reason is because, as I’ve hopefully convinced you, animals (including humans!) are awesome, and we want to figure out how and why they do the bizarre and amazing things they do. Phew, that was exhausting, but I hope you found it worthwhile! I learned so much about sensory ecology in this class and hopefully passed some of that on to you. Recently, I was lucky enough to participate in Lund University's Sensory Ecology Course. This international post-graduate course is held from late September through early October on even years only (i.e. next course is fall 2016). The course recruits experts from around the world, each specializing in a different sensory modality (e.g. vision, olfaction, audition, electro-sensing, etc.) to train eager young scientists in the field of sensory ecology. I think I can speak for others in the class by saying, we learned a great deal and made contacts that will last a lifetime, and were lucky enough to be hosted by the world-renowned and extremely hospitable Lund Vision Group. My labmate, Brian Hoover, actually gave me the idea for this post at the end of the course. So, in no particular order, here are the 10 most amazing things I learned. I will write about five this week, and the other five next week. 10. Why do giant squid have giant eyes? Can you guess which animal has the largest eye? The title of this bullet point gives it away but, it’s not an elephant or even a whale, it’s actually a giant squid. Measuring nearly 30 centimeters across with a pupil 9 centimeters diameter, giant squid eyes are roughly three times larger than the next largest eye in the animal kingdom (that of the Swordfish). Considering eyes are one of the most metabolically expensive structures to produce and maintain, why have eyes so large? Is it to find their prey in the deep ocean? To find a mate in the dim light? Nope! Scientists believe that giant squid have such massive eyes to be able to see the faint bioluminescence of meso and bathypelagic plankton, which light up when they are disturbed by a passing Sperm Whale, the squid’s main predator. It appears the giant eyes evolved to try and help the squid avoid becoming a giant serving of calamari! Read the article here. 9. Bats “see” with sound One thing impressed upon me by Annemarie Surlykke is that bats, which often get publicized as blood-drinking disease vectors, are finely-tuned auditory machines. To locate prey, they use echolocation, producing a series of loud (up to 140dB at the source; for reference, a jet airliner taking off 25 meters away is ~150dB) high-frequency (up to 20-80 kHz; humans can hear up to ~20kHZ) clicks and the returning echoes to locate prey or navigate in total darkness. Insectivorous bats’ hearing is so sensitive that they can use the Doppler shift generated by the returning echoes from beating insect wings to differentiate potential prey items from inanimate objects, such as buildings, trees, and leaves. Perhaps most amazingly, bats can alter the qualities of their echolocation clicks situationally. For example, by adjusting their mouth opening, a bat can alter the frequency and directionality of echolocation clicks, actively changing both while they hunt. Searching bats produce directional, evenly-spaced (temporally), high-intensity (dB) clicks. Once a bat has located a potential prey item, it clicks more frequently to increase the amount of detailed information about the target, then captures the item by widening its echolocation beam and reducing the intensity (dB) of it’s echolocation clicks to reduce far off echoes from distracting the bat from its target. Watch it in slow motion here. Unlike humans that have static vision (for example, we can’t widen our field of view or adjust how far away we can see), bats have dynamic auditory “vision” by being able to alter the temporal spacing, intensity, frequency, and beam-width of their echolocation clicks. So think twice the next time you utter the phrase “blind as a bat”. Check out Annemarie's Lab Website for more information. 8. A brief history of vision According to vision expert, Dan-Eric Nilsson, organisms’ eyes are just about as good as it can get in terms of their physical properties. Advanced eyes with a camera-type lens (like ours) and a highly specialized region of the retina for detailed photon reception (in our case, the fovea) can produce images as sharp or sharper than the best cameras or machines ever produced. If vision is to improve over future evolutionary time, it is likely the advancements will be made in neurological image processing (i.e. transduction), rather than physical eye-design. What’s more, the evolution of vision was likely a very punctuated event. Meaning that the development of the first visual pigments and the ability for single-celled organisms to detect and respond to ambient light conditions (the simplest form of vision), all the way to complex, image forming, camera lens-type eyes occurred during a fairly discrete time period, beginning approximately 800 million years ago and concluding around the end of the Cambrian Explosion, roughly 500 million years ago. There has been limited development of vision, relatively speaking, in times before or since that ~300 million year period. Considering there has been life on earth for ~3.5 billion years, the fact that the majority of visual development happened over a < 10% period of that time is pretty astonishing.
6. Swimming with an internal compass After hatching out of their eggs on beaches in the southeastern United States, young Loggerhead Sea Turtles (Caretta caretta) undertake a massive journey over the next 5-10 years of their lives, circling around the entire North Atlantic Gyre. How do they make it around this visually-featureless landscape back to the same area where they took their first strokes in the ocean nearly a decade earlier? Well, Ken Lohmann and colleagues discovered that one way these turtles navigate on such enormous spatial scales is via the earth’s magnetic field. These turtles can use the varying magnetic field strength and inclination angle to figure out which direction to swim. By systematically removing all other sensory cues, it has been shown experimentally that if young Loggerhead Turtles are magnetically displaced, they tend to swim in the direction they would need to swim to stay within the North Atlantic Gyre, thus keeping them on their proper migratory trajectory. The exact mechanism by which these turtles (or any animal known to use a magnetic compass) detect and orient to magnetic fields remains unknown. Stay tuned next week for amazing facts #s 5-1!  Holding an experimental perch at the Bodega Marine lab earlier this summer. Holding an experimental perch at the Bodega Marine lab earlier this summer. My research on marine plastic debris is featured in the most-recent bulletin of the American Fisheries Society (Davis' subunit), put together by my good friend and superb sturgeon-surgeon, Emily Miller. Check out the bulletin describing the work (and other fish-related research going on at UC Davis) here. And if you're curious, stay tuned for more results from my summer's research coming up soon! After just coming back from the Graduate Group in Ecology’s (GGE) White Mountain Odyssey for the fourth time in five years, I wanted to write this post as a cathartic decompression of sorts. Also, since this was the last Odyssey I (as well as most people in my 2010 cohort) will help lead, we need replacements! If you are not part of the GGE and wondering what the heck the Odyssey is, let me give you the cliff notes description: it's a week-long trip through the mountains of eastern California put on by the GGE with the purpose of getting the incoming cohort of graduate students familiarized with several UC Reserves and their fellow GGEers, specifically their new cohort-mates. The trip is done with the new students packed in several (4-6, depending on the size of the incoming cohort) large vans, with two current GGE students responsible for each van. These current GGE students who drive the vans are deemed “TAs,” even though this is nothing like any other TA experience on earth. If you enjoyed your Odyssey and are thinking about TAing for the first time, be prepared to have twice as much fun as you had on your own Odyssey, despite being twice as tired at the end. Here are five reasons why I think every GGE student, whether you’re a second-year or a seventh-year, should consider TAing the Odyssey. 1) Fosters a strong connection to the new cohort Just as on your own Odyssey, interpersonal bonds form quickly and deeply between people who were complete strangers only a week before – throwing 30-50 people into a crazy situation together has the tendency to do that. The one year I didn’t attend the full Odyssey (2013) since I’ve been at UC Davis, I felt disconnected from the then first-years (currently second-years) when they returned from the trip. It was a depressing feeling for me. I would meet them at seminars or social gatherings over the following year where we would talk, but the conversations were often brief and superficial – the “where are you from?” “what lab are you in?” “what are you planning to study?” type questions – as conversations in those situations usually are. TAing the Odyssey is one surefire way to get to know the new cohort quickly and well, before their time in Davis even begins. 2) Fosters a strong connection to the older cohorts The bond between TAs forms well before the Odyssey departs Davis, often months before. This previous year, for example, James and I would spend nights several months in advance planning how to best implement our van plan. The connection you form with you co-van driver will probably be the strongest, but I actually found the connections formed between TAs who are not in the same van to be the most valuable. I have made new friends and become much closer with people I have TAed Odysseys with, especially those driving other vans. At minimum, TAs on the same Odyssey will share jokes and stories that last a lifetime. 3) Fosters a strong connection and appreciation for GGE faculty and administration Even not including the awesome students, the GGE is such a wonderful community to interact with; it can be easy to forget that if you're too bogged down in your own research. On the trip, we have no fewer than six professors/researchers tagging along for at least part of a day. As a TA, you have unfettered access to these individuals if you so desire. Also, it’s a great way to kick back and have a relaxing conversation with the Student Affairs Officer (SAO; currently Holly Hatfield, formerly Silvia Hillyer), who spends most of her work-year helping you navigate the bureaucracy and administration of a large university with ease. Spending quality time with the SAO on the Odyssey is not only fun, but may make it less uncomfortable the next time you need to ask her for help. 4) TAs just wanna have fun I find graduate students to be generally plagued with guilt when not working on their research. The Odyssey is a rare opportunity when you can forget about that pesky analysis that hasn’t been working, let loose, and have fun for a week, while still being able to say it’s all in the name of service to your graduate group. Besides to have a great time, the other main reason I have TAed the Odyssey three times is to "pay it forward," to give the incoming students that overwhelmingly positive first experience in the GGE that was once given to me.  At 14,252 feet, White Mountain Peak (on a beautiful day in 2014), which we climb at the end of the trip, is one of the tallest mountains in the lower 48 states. At 14,252 feet, White Mountain Peak (on a beautiful day in 2014), which we climb at the end of the trip, is one of the tallest mountains in the lower 48 states. 5) Getting different experiences revisiting places you rarely see Since I do marine research, I cherish going on the Odyssey because I get to visit several gorgeous regions in California that I rarely or never visit otherwise. Visiting the White Mountain moonscape is probably what I look forward to the most, but stunning Mono Lake as well as the high peaks of the southern Sierra Nevada are also up there for me. Every year, the flora and fauna you get to see is a bit different, as is the weather. On White Mountain Peak for example, I’ve seen a blustery snowstorm (in 2011) and a day warm and calm enough to wear t-shirts and play cards at the summit (in 2014). Moreover, since the group changes, this gives visiting each location a unique vibe every year. Addressing detractions:
1) “I’m just not that social or high-energy” This is the most common excuse I hear from people who have considered TAing the Odyssey, but ultimately decide against it. The idea that only the most social, high-energy GGE students should TA the Odyssey is misguided. The trip actively needs more mellow TAs to balance out the consistently high-energy ones. The low key first-years may even seek out the mellower TAs as a refuge. The mix of hyper-social and more reticent TAs is crucial for maintaining balance on the Odyssey. 2) “I can’t take time off during the Odyssey” This reason I unfortunately cannot dispute. Some of us have field and/or lab work that is phenologically-constrained and are therefore unable to take any time off in the late summer/early fall. For those people, I am truly sorry, because you are missing out on a wonderful experience. Luckily, for most of us, we can take the time off (5 work days), the question is: will we? Conclusion: YOU should volunteer to TA the White Mountain Odyssey!  Sperm Whales defecating on the ocean surface. Picture by Tony Wu. Sperm Whales defecating on the ocean surface. Picture by Tony Wu. Two weeks ago, Deep Sea News had a fantastic post summarizing all the recent work done on marine nutrient recycling via excretion. Believe it or not, this research topic is really gaining steam (I feel like I missed a poop-joke in there somewhere)! Read their write up of the recently-published research here.  Evan Eskew presenting on disease ecology at Folsom Lake College. Evan Eskew presenting on disease ecology at Folsom Lake College. Two weeks ago on Earth Day, I was honored to be part of a very special event. Dr. Steven Holzberg (a professor of biology at Folsom Lake College) and I finally saw our brainchild come to fruition: a free, public symposium on the ecological effects of global anthropogenic change. Considering how difficult it can be to get the general public interested in science during their free time, I feel as though the event was a great success. Students, parents, emeritus professors, and other members of the general public came out to Folsom Lake College with open ears and open minds. Unlike most scientific presentations, there was no professional glory or payment of any kind for my fellow grad student presenters and me, but rather, the event was fueled by our desire to communicate science to a broad audience.  Jason Riggio presenting on habitat fragmentation at Folsom Lake College. Jason Riggio presenting on habitat fragmentation at Folsom Lake College. I must say I was surprised when every graduate student I initially asked to help with this event responded so positively. However, I was disappointed to be so surprised by their gung-ho attitudes because I would have expected quite the opposite. In my less-than-a-decade in science, I have noticed that oftentimes the more high-profile one's scientific research becomes, the less the researcher sees the need to communicate it to those outside his/her field. Basically, scientists frequently have trouble seeing the forest for the trees. Recently, I even shared these feelings with a UC Davis professor who responded with his/her hypothesis that “there is an inverse relationship between how big your name is in science circles and how well you present a talk.” I would have to agree, and that realization has been one of the most disheartening ones I've had in graduate school thus far. That is not to say that there haven’t been some very effective and successful science communicators in recent history. Over the last half-century alone, I can think of a handful of scientists who made science communication as, if not more, important than their science itself. For example, my childhood love for science was partially sculpted by watching Bill Nye the Science Guy on PBS. Harvard biologist, E.O. Wilson has won not one, but two Pulitzer Prizes for his non-technical science writing. Over the past three decades, Jane Goodall has written over twenty popular and children's science books about the great apes, our species’ closest living relatives. From BBC series such as The Life of Birds and Planet Earth, David Attenborough has become a household name. And fellow New York City native Neil DeGrasse Tyson continues to blow my mind every week with his re-working of Carl Sagan’s classic popular science TV series, Cosmos (watch every full episode here for free). Considering all that, what does it mean to be a scientist anyway? In the modern age, where communication is instantaneous and necessary for transferring ideas, I feel as though part of being a great scientist should include being a superb (not just an "OK") communicator. Whether that's through writings, presentations, or both, communicating scientific discoveries to a broader audience (scientific and otherwise) is as critical as the discovery itself.  Sarah Jennings and I at Patwin Elementary School in Davis, CA. Sarah Jennings and I at Patwin Elementary School in Davis, CA. There's certainly a place in science for individuals who devise incredibly complicated laws and techniques, using these to generate discoveries within science, but all the while are totally helpless communicating their findings to anyone outside their highly-specialized sub-discipline. I think of those people as "brilliant" or "genius," and would say that those types don't even fit in to my holistic definition of what a scientist should be. In my view, a scientific genius can be a scientist, but s/he doesn't have to be. Said another way, a scientist can be brilliant, but s/he doesn't have to be. Being an effective communicator is an essential component of what a scientist is, in my mind. And so, I feel it is fitting on National Teacher’s Day to make a charge to all my friends and colleagues working in science to embrace the teacher in you and display your science to the world. What’s the worst that can happen? You may even inspire a great future scientist in doing so. Just yesterday, I heard a brilliant idea: economically incentivize people to clean up plastic waste from the world's beaches, oceans, and waterways. The Canadian-based organization that is attempting to do this is called Plastic Bank. According to co-founder Davis Katz, plastic is more valuable than steel by weight and with literally billions of tons of plastics littering the world's beaches and waterways, there's trillions of dollars worth of plastic refuse out there just waiting to be recycled! This is not the first time that economists have tried to mitigate a global environmental problem using a monetary solution. In fact, when I first heard of what Plastic Bank is trying to do, it made me think of carbon taxing, which has been implemented around the world with varying results. With that being said, I have one word for you: plastics. Are you intrigued by this market-based solution to a global environmental problem? Then follow Plastic Bank on twitter and like their facebook page!
The mini-symposium ran by eight awesome UC Davis Ecology PhD students and hosted by Folsom Lake College yesterday for Earth Day was a rousing success! Thanks to everyone who helped make it happen especially my fellow presenters and Dr. Steven Holzberg of Folsom Lake College. I will have a longer post about the event next week, but for now, just know that the organizers and presenters had a great time! Looking for something to do on the evening of Earth Day 2014?
Well, you're in luck because eight Ecology graduate students at UC Davis have teamed up with the Biology faculty at Folsom Lake College to run a FREE, public mini-symposium on the Ecological Effects of Global Anthropogenic Change. Topics such as climate change, habitat fragmentation, invasive species, over-harvesting, and more will be discussed. The symposium (including panel discussion) will run from 4:00-7:30pm, next Tuesday (4/22). See the program flyer for more details. We hope to see you there! 3/6/2014 Can DMS facilitate a tritrophic mutualism between primary producers and top predators?Read NowAfter roughly two years in the making, the first chapter of my PhD is in print, with full open access! This post summarizes that study. A mutualism is a biological relationship where both parties benefit from the interaction. A common example is the relationship between Remora fish and sharks. The predatory shark can simply turn around and eat the Remora, but it doesn’t. Why? The reason is because the Remora provides the shark a service: it cleans ectoparasites off the shark’s body; in exchange, the shark gives the Remora a free meal and transport wherever the shark goes. That type of mutually beneficial relationship is what we have investigated in this paper. Complicating matters, we deem the mutualism in our paper a “tritrophic mutualism,” so what do we mean by “tritrophic”? A tritrophic interaction is one where two organisms, separated by at least one trophic level, interact in some biologically meaningful way. A common example of a tritrophic interaction is between lima bean plants and predatory insects. Herbivorous spider mites consume lima bean plants. As a result of being grazed, these lima bean plants release airborne compounds that alert predatory insects the plants are being attacked (Dicke 1986, Dicke et al. 2003). Olfactory-searching predatory insects detect odors emitted from damaged lima bean plants and use these odors to find the herbivorous spider mites, which are the preferred prey for many of these predatory insects. The interaction between lima bean plants (primary producer) and olfactory-searching predatory insects (secondary consumer) is the tritrophic interaction. This is precisely the type of relationship we investigated in the marine environment between phytoplankton (primary producers) and procellariiform seabirds (top predators). To support this, we used diet data from 18 species of procellariiform seabirds collected over ~50 years. Dimethyl sulfide (DMS) is produced when phytoplankton cells burst, often due to predation, and DMS is known to be attractive to some species of procellariiform seabirds, but not all. We preformed a meta-analysis to show that those species of procellariiform seabirds that are attracted to DMS specialize in herbivorous crustacean prey. In other words, those seabird species attracted to DMS are using the phytoplankton-derived compound as an olfactory cue to find their preferred prey. However, this alone isn’t enough to claim the interaction between phytoplankton and seabirds is mutualistic; we still needed to demonstrate a possible benefit the phytoplankton would receive from the attracted seabirds. One obvious answer is predatory release: seabirds are attracted to DMS, find the odor source, and depredate the herbivorous crustacea thereby “releasing” the phytoplankton from grazing pressure. While this is likely happening on some spatial scale, we instead chose to focus on the possible benefit foraging seabirds may provide phytoplankton through trace nutrient recycling via their defecation. In the Southern Ocean, soluble iron limits primary production, and because iron is toxic to vertebrates if sequestered at high levels, most of ingested iron is excreted. It has recently been shown that whale and seabird feces are very high in iron content, relative to Southern Ocean seawater. Iron enrichment via defecation by top predators may therefore enhance primary productivity in portions of the iron-limited Southern Ocean. Perhaps surprisingly, this is not the first time the concept of top predators fertilizing phytoplankton via their excrement has been considered. This concept has been tested recently with respect to whales in iron-limited (Lavery et al. 2010, Nicol et al. 2010) and nitrogen-limited (Roman and McCarthy 2010) systems. What makes our study unique is that we provide a mechanism – the chemoattraction of procellariiform seabirds to phytoplankton-produced DMS – linking the whole system together. It is possible that whales also use DMS as a foraging cue, although no data exists on the subject.
In summary, I’ll quote the last sentence of our paper, “results presented here illustrate a fundamental, albeit understudied, link between apex predators and the base of the pelagic food web, suggesting that a decline in seabird populations could negatively affect overall marine productivity.” (Savoca and Nevitt 2014). Last weekend, I had the pleasure of presenting my research on marine plastic debris at the 7th Annual Ecology Graduate Student Symposium. Attendees included over 100 undergraduate and graduate students, professors from at least three different institutions, and members of the general public. It was a great day filled with interdisciplinary science, beautiful artwork, and stunning photographs. The symposium even got covered by the UC Davis newspaper, The California Aggie. Read the story here. To top it off, I won the award for best talk! Considering the research I do on plastic debris is rather unusual, it was exciting to get a positive reception and encouraging feedback, especially since it was the first presentation I’ve given on this work.
Now I’m off to Honolulu, Hawaii for the 17th Ocean Sciences Meeting where I will present my preliminary findings to a much larger scientific audience. Whatever happens at the conference in Hawaii, I will be better off having presented at the EGSA Symposium first. Big thanks to James Farlin, Grace Ha, Katie Eskra, Matt Whalen, and many other volunteers too numerous to list for organizing such a fantastic event. Visit UC Davis’ Graduate Group in Ecology’s website for more information on this powerhouse program. Also, check out the Ecology Graduate Student Association’s webpage to learn about some of the great events and amazing work produced by members of my graduate group! 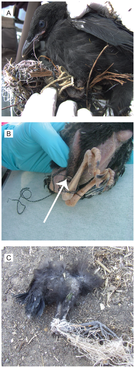 Great work by a good friend and colleague Andrea Townsend, demonstrating the negative impacts plastic debris has on a common land bird, the American Crow. With so much research on plastic debris in marine ecosystems, it's good to see terrestrial communities garnering some attention. About the photo: American Crow nestlings in Yolo County, California found tangled in plastic debris in their nests. Figure taken from Townsend and Barker 2014. Read the full article here. Heard back from the Society for Integrative and Comparative Biology (SICB) awards committee today and I am happy to announce I was selected as one of the 28 winners this year. First direct funding for my studies of marine plastic debris!
Find out more about SICB graduate student support options. Olfaction simply refers to the sense of smell. The chemosensory/olfactory system is the most ancient sensory system that we know of. Nevertheless, it is perhaps the least-studied, inciting much less scientific interest than vision or hearing, for example. There are many possible reasons why this disparity exists. One of the most compelling reasons I’ve heard is simply because humans have relatively poor olfactory abilities. Humans are primarily visual and auditory creatures, every other sense comes after. For other animals however, this may not be the case. While dogs have fine vision (despite being red-green colorblind) and exceptional hearing, they are also fantastic smellers. For many fish that inhabit murky waters, their sense of smell is how they perceive their world. Even for fish that are visual predators, like salmon, they still need to use their world-class noses to find their home stream where they will eventually reproduce and die.  Turkey Vultures at an evening roost. Turkey Vultures at an evening roost. The study of olfaction in birds has a long and confusing history. Until the 20th century, it was believed that birds did not have a sense of smell at all. Classic studies conducted in the 1820s by John James Audubon on the Turkey Vulture (Cathartes aura) are some of the first known experiments on avian olfaction. Surprisingly, Audubon’s conclusion was that these vultures have no sense of smell. We now know this not to be true. In fact, a Turkey Vulture’s nose is so attuned to methyl mercaptan – a compound found in decaying flesh as well as crude oil extract – that early 20th century engineers would use the presence of these birds to detect leaks in oil pipelines well before humans could. While it is true that many birds are primarily visual animals and may have poor senses of smell, we don’t have very much data on the subject. We do know, however, that new world vultures, kiwis (the flightless birds from New Zealand, not the fruit), and tube-nosed (a.k.a. procellariiform) seabirds lead the pack, having the best avian senses of smell. Not only that, but these birds have some of the most sensitive olfactory systems known in the entire animal kingdom. My lab studies olfaction using procellariiform seabirds as model organisms. After two decades of research, Gabrielle Nevitt and her collaborators have discovered some amazing things about these “noses with wings”. It had been well known for decades that procellariiform seabirds had an excellent sense of smell from anatomical (Bang 1966) and behavioral studies (Grubb 1972, 1973), but Nevitt was able to show how specific food-related odors attract different species of seabirds, depending on what they prefer to eat. Her results have shown that seabird species feeding on primary consumers (e.g. krill, copepods, etc.) are often attracted to the phytoplankton-derived odorant dimethyl sulfide (DMS; Nevitt et al. 1995), while seabird species foraging higher on the food chain (e.g. predatory fish and squid) tend to be attracted to krill-related odors, such as tri-methyl amine and tri-methyl pyrazine (Nevitt 1999, Nevitt et al. 2004). Furthermore, it was discovered that these birds can detect specific airborne odors in parts per billion concentrations or lower, which is at least three orders of magnitude (1000x) more sensitive than our noses are for the same odors!  Shoulder deep in a Storm-Petrel burrow on Bon Portage Island. Shoulder deep in a Storm-Petrel burrow on Bon Portage Island. In addition to finding food, these birds have also been shown to use their sense of smell in the contexts of homing and individual recognition. In what many may come as a shock to some, many of the smaller species of procellariiforms actually nest underground in earthen burrows that can be up to a meter long. It’s incredible to imagine these little birds (most of the burrow-nesting seabirds are smaller than a crow) excavating their own burrows! For these burrow-nesting procellariiforms, they are often active at night, returning or departing from their colonies in pitch blackness. It has now been shown that burrow-nesting Blue Petrels (Halobaena caerulea), Common (Pelecanoides urinatrix) and South-Georgian Diving Petrels (P. georgicus), as well as Thin-billed (Pachyptila belcheri) and Antarctic Prions (P. desolata) use smell to locate their burrows in complete darkness (Bonadonna et al. 2003a, Bonadonna et al. 2003b, Bonadonna et al. 2003c). In another simple, yet elegant, experiment Bonadonna and Nevitt (2004) demonstrated that Antarctic Prions were able to recognize and distinguish their personal odor from the odor of their partner as well as a randomly selected prion. It was found that these birds actually preferred the scent of their partner’s odor to their own, and of the three different odors, they were least interested in the odor of a randomly-selected conspecific. As always in science, answers only lead to more questions. The lab I work in is currently involved in a multi-year project studying Leach's Storm-Petrels (Oceanodroma leucorhoa) to determine if these diminutive, burrow-nesting seabirds select mates by scent with a particular MHC, a complex cluster of ultra-variable genes involved in immune function. Many more questions related to olfaction in wild animals can and should be addressed; there’s a whole world of discovery out there, who nose what one may find next!  Plastic-covered Hawaiian Island moves toward possible superfund designation. A sad, but positive step for Tern Island. Read the full story. |
Details
AuthorMatthew Savoca holds a PhD in Ecology from the University of California, Davis. His research interests include sensory behavioral ecology, marine conservation biology, and seabird ecology. Categories
All
Archives
December 2021
|






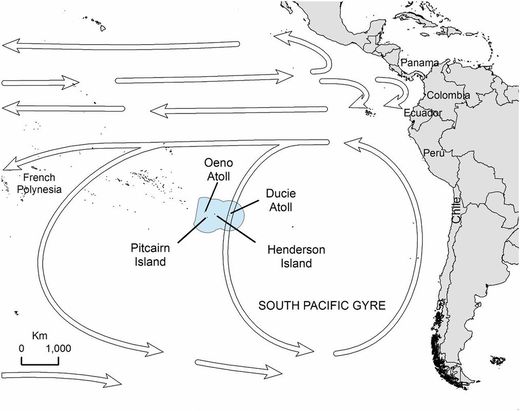











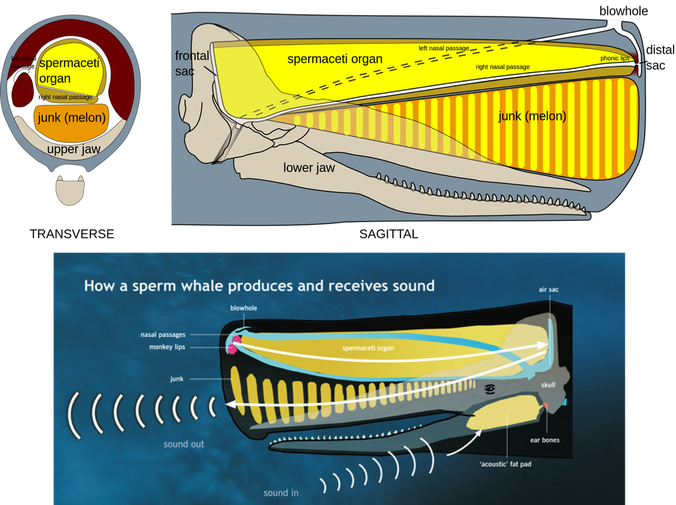




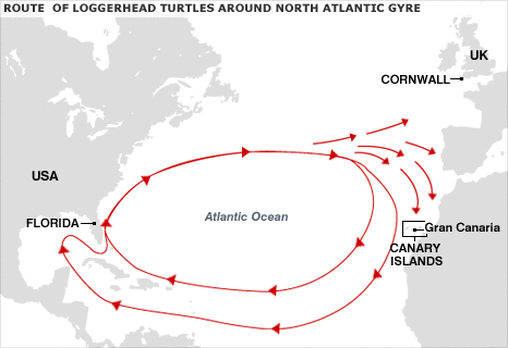





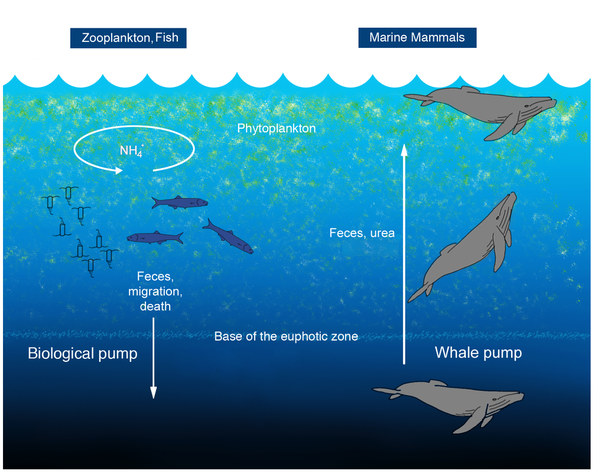

 RSS Feed
RSS Feed
