|
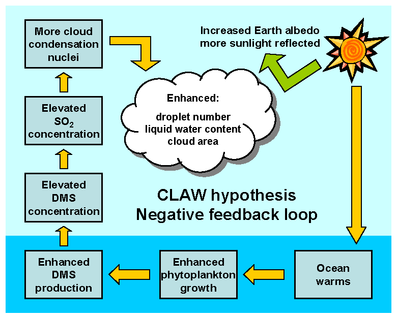
10/21/2013 The tale of dimethyl sulfide: A global climate regulator turned marine infochemicalRead NowI must admit, I took the idea for the title of this post from a recently-published paper by my PhD advisor (Nevitt 2011. Integ. & Comp. Biol. 51:819-825). This post is about how a simple, plant-derived molecule influences global climate and mediates biotic interactions throughout the marine food web. The molecule in question is dimethyl sulfide (DMS) and it is so important that many scientists have dedicated their entire careers studying it in action. DMS consists of a single sulfur molecule stuck between two methyl (CH3) groups. Produced by marine and freshwater algae, DMS is a breakdown product of dimethylsulfoniopropionate (DMSP; an osmoregulatory compound) and forms when algal cells lyse. DMS has a very low boiling point, which means that once it’s formed in the water, it readily evaporates. For a long time, DMS was thought to be nothing more than a biological waste product of DMSP catabolism. That all changed in the late 1980s, when a paper was published in Nature titled: Oceanic Phytoplankton, Atmospheric Sulfur, Cloud Albedo and Climate (Charlson et al. 1987). That article has since been cited over 2,100 times according to the Web of Science. This paper was revolutionary because it suggested that DMS plays a significant role in global climate regulation. When DMS evaporates, it oxidizes to form sulfur dioxide, which is an important cloud-forming molecule. An increase in the amount of algae in the ocean would theoretically increase the concentration of DMS in the air and subsequently form more clouds. Clouds do a great job of reflecting incident solar radiation back into space. This increase in the earth’s reflectance (termed “albedo”) would lower global temperatures. The CLAW hypothesis, as this theory is now known, is an acronym for each of the authors’ last names of the original paper (Charlson et al. 1987). DMS wasn’t well known among zoologists until the mid-1990s when it was first suggested that procellariiform seabirds (i.e. petrels and their allies) use the scented compound to help locate biologically-productive regions to feed (Nevitt et al. 1995. Nature. 376:680-682). Since then, it has been shown that organisms as diverse as penguins, reef fish, seals, and whale sharks are attracted to DMS/P in foraging contexts. A biochemical that provides information to another organism is known as an infochemical. The concept that a plant-derived compound signals to attract predators has been known for decades in the plant-insect literature, but this idea is fairly new in the marine realm.
That is where my work comes in. In addition to studying plastic ingestion in procellariiform seabirds, I am also investigating the role these amazing birds play in a marine tritrophic interaction, mediated by the infochemical, DMS. From its humble beginnings as a mere biological waste product, DMS has turned into one of the most well-studied molecules on the planet. An interesting tale, indeed.
1 Comment
Isabel Geffenr
10/22/2013 08:36:28 am
This sounds like it could be game-changing in our understanding of climate change. WOW!
Reply
Leave a Reply. |
Details
AuthorMatthew Savoca holds a PhD in Ecology from the University of California, Davis. His research interests include sensory behavioral ecology, marine conservation biology, and seabird ecology. Categories
All
Archives
December 2021
|
 RSS Feed
RSS Feed
